Exosomes in Aesthetic Medicine: Science, Manufacturing, and Quality Considerations
Introduction
In recent years, exosome-based therapies have emerged as one of the most promising advancements in regenerative and aesthetic medicine. From skin rejuvenation to hair restoration, exosomes are redefining how clinicians approach cellular repair and tissue regeneration.
However, as the market expands rapidly, a critical challenge arises:
Not all exosome products are created equal.
Understanding what exosomes are, how they function, and how they are manufactured is essential for making informed clinical and commercial decisions.

What Are Exosomes?
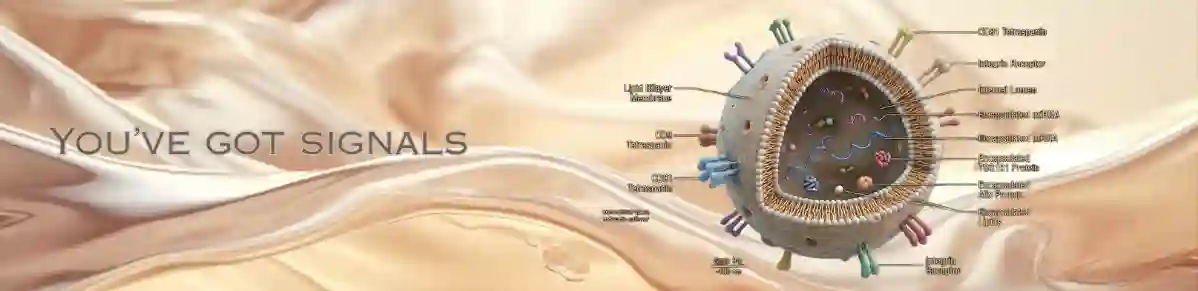
Exosomes are membrane-bound extracellular vesicles (EVs), typically ranging from 30 to 150 nanometers in diameter, originating from the endosomal compartment of eukaryotic cells.
Their formation is a tightly regulated intracellular process involving the endosomal sorting pathway:
- Endocytosis leads to the formation of early endosomes
- These mature into late endosomes, also known as multivesicular bodies (MVBs)
- Within MVBs, intraluminal vesicles (ILVs) are formed through inward budding of the endosomal membrane
- When MVBs fuse with the plasma membrane, ILVs are released extracellularly as exosomes
This biogenesis is mediated by complex molecular machinery, including:
- ESCRT (Endosomal Sorting Complex Required for Transport) proteins
- Tetraspanins (CD9, CD63, CD81), which are commonly used as exosomal markers
- Lipid-dependent mechanisms involving ceramide and sphingolipid pathways
Molecular Composition
Exosomes are not inert vesicles, they are biologically active nanocarriers with a highly organized structure.
Lipid Bilayer Membrane
The exosomal membrane is enriched with:
- Cholesterol
- Sphingomyelin
- Phosphatidylserine
This unique lipid composition:
- Provides structural stability
- Protects internal cargo from enzymatic degradation
- Facilitates membrane fusion with recipient cells

Protein Cargo
Exosomes carry a selective set of proteins reflecting their cell of origin:
- Surface proteins
- Tetraspanins (CD9, CD63, CD81)
- Integrins (cell targeting and adhesion)
- MHC molecules (immune modulation)
- Cytosolic proteins
- Heat shock proteins (HSP70, HSP90)
- Enzymes involved in metabolism
- Signal transduction molecules
- Functional growth factors and cytokines
- TGF-β
- VEGF
- FGF
- EGF
These proteins contribute directly to cell signaling, angiogenesis, and tissue remodeling.

Nucleic Acid Cargo
One of the most critical functional aspects of exosomes is their genetic payload:
- mRNA → can be translated into functional proteins in recipient cells
- microRNA (miRNA) → regulates gene expression post-transcriptionally
- Long non-coding RNA (lncRNA) → modulates epigenetic and transcriptional processes
For example:
- miRNAs can suppress pro-inflammatory pathways
- Or activate fibroblast proliferation and collagen synthesis
This makes exosomes a form of horizontal gene regulation system between cells.

Biological Role: Intercellular Communication
Exosomes function through a biological signaling mechanism, not as simple topical ingredients. At the molecular level, exosomes function as targeted delivery systems.
They interact with recipient cells via:
- Receptor–ligand binding
- Endocytosis
- Direct membrane fusion
Once internalized, their cargo:
- Alters gene expression
- Modulates signaling pathways
- Reprograms cellular behavior
In dermatological and aesthetic applications, this leads to:
- Activation of fibroblasts → collagen and elastin synthesis
- Modulation of keratinocyte proliferation and differentiation
- Regulation of inflammatory cascades (e.g., NF-κB inhibition)
- Stimulation of hair follicle stem cells via Wnt/β-catenin signaling
Unlike traditional treatments, exosomes do not replace cells, they instruct existing cells to perform better.

Functional Specificity
It is important to emphasize that exosomes are not uniform entities.
Their biological activity depends on:
- The cell source (e.g., MSC vs fibroblast)
- The physiological state of the parent cell
- The microenvironment during production
This results in significant variability in:
- Cargo composition
- Signaling potency
- Clinical outcomes
How Are Exosomes Manufactured?
The production of exosomes is a highly sensitive and technically demanding process.
1. Source Selection
Exosomes are derived from cultured cells, commonly:
- Mesenchymal stem cells (MSCs)
- Fibroblasts
- Other specialized cell lines
The origin and quality of the parent cells directly influence the final product.
2. Cell Culture Conditions
Cells are grown under controlled laboratory environments:
- Serum-free or defined media
- Strict contamination control
- Optimized oxygen and nutrient levels
Even minor variations in culture conditions can significantly alter:
- Exosome yield
- Biological activity
3. Isolation and Purification
This is one of the most critical steps.
Common methods include:
- Ultracentrifugation
- Filtration
- Chromatography
High-quality manufacturing ensures:
- Removal of impurities (proteins, cell debris)
- Preservation of vesicle integrity
Poor purification leads to diluted or contaminated products.
4. Characterization and Quality Control
Advanced manufacturers perform:
- Particle size analysis (NTA)
- Protein marker validation (CD9, CD63, CD81)
- Sterility and endotoxin testing
This step ensures consistency, safety, and efficacy.
5. Stabilization and Formulation
Exosomes are highly sensitive.
To maintain stability, manufacturers may use:
- Lyophilization (freeze-drying)
- Specialized buffers or carriers
Improper stabilization can destroy:
- Membrane integrity
- Biological activity

Why Do Exosome Products Have Different Prices?
One of the most confusing aspects for clinicians and distributors is the wide price variation in the market.
This variation is not arbitrary, it reflects fundamental differences in quality, process, and integrity.
How Manufacturers Reduce Costs (and What It Means)
To compete in a growing market, some manufacturers reduce production costs. This can be achieved through:
- Using lower-grade or undefined cell sources
- Simplifying purification processes
- Skipping advanced characterization tests
- Reducing exosome concentration
- Compromising on storage and stabilization methods
While these strategies lower the price, they may lead to:
- Reduced biological activity
- Inconsistent clinical outcomes
- Shorter shelf life
- Increased risk of impurities
1. Source Material Quality
One of the most critical—yet often overlooked—determinants of exosome quality is the source of the parent cells. At the molecular level, exosomes are a reflection of the cell they originate from, both in composition and biological function.
This means that the therapeutic profile of an exosome product is inherently tied to its cellular origin.
- High-quality, well-characterized cell lines are expensive
- Poor-quality or undefined sources reduce cost—but also reliability
2. Manufacturing Standards
The manufacturing environment plays a decisive role in determining the safety, consistency, and biological activity of exosome products.
GMP-Certified Facilities
Production in Good Manufacturing Practice (GMP)-certified facilities requires substantial investment in infrastructure, systems, and regulatory compliance.
This includes:
- Controlled cleanroom environments (ISO-classified)
- Strict regulation of air quality, particulate count, temperature, and humidity
- Minimization of microbial and endotoxin contamination
- Standardized and validated processes
- Reproducible cell culture conditions
- Defined protocols for isolation, purification, and storage
- Batch-to-batch consistency ensured through validated SOPs
- Comprehensive quality control testing
- Sterility testing
- Endotoxin levels (e.g., LAL assay)
- Particle characterization (size distribution, concentration)
- Marker validation (CD9, CD63, CD81)
- Traceability and documentation
- Full tracking from cell source to final product
- Batch records, deviation reports, and release criteria
The result is a product with:
- High consistency
- Predictable biological performance
- Clinical reliability
Non-Standard or Research-Grade Production
In contrast, some products are manufactured in non-GMP or research-grade laboratory environments, where cost reduction is prioritized.
This may involve:
- Less controlled environments
- Higher risk of contamination (microbial, protein impurities)
- Variability in environmental conditions affecting cell behavior
- Lack of process standardization
- Differences in culture media, timing, and harvesting methods
- Inconsistent purification techniques
- Limited or absent quality testing
- No standardized measurement of exosome concentration
- Incomplete validation of biological markers
- Minimal sterility or endotoxin control
- Reduced traceability
- Limited documentation of cell origin and production history
While these approaches reduce production costs, they often lead to:
- Batch variability
- Unpredictable clinical outcomes
- Lower biological potency
In exosome-based therapies, manufacturing standards are not merely regulatory requirements—they are directly linked to biological function and clinical outcome. Two products may appear similar in labeling, yet differ significantly in Purity, Potency, Safety profile.
3. Purity and Concentration
Beyond the source and manufacturing environment, the actual therapeutic value of an exosome product is largely defined by two measurable parameters:
- Purity → What proportion of the product is truly functional exosomes
- Concentration → How many exosome particles are delivered per unit volume
These two factors directly determine both biological performance and production cost.
High Concentration of Functional Exosomes
High-quality products are characterized by:
- Quantified particle concentration (e.g., particles per mL via NTA analysis)
- A high proportion of intact, biologically active vesicles
From a manufacturing standpoint, achieving this requires:
- Large-scale cell culture systems
- More cells → more exosome yield
- Higher media consumption and longer production cycles
- Efficient isolation techniques
- Ultracentrifugation, tangential flow filtration, or chromatography
- Multiple processing steps to concentrate vesicles without damaging them
- Minimization of loss during purification
- Exosomes are fragile; aggressive processing can destroy them
- High-end processes aim to preserve both quantity and functionality
Cost implication:
- Higher raw material usage
- Longer processing time
- Advanced equipment and skilled operation
Clinical implication:
- Stronger signaling effect
- More predictable and reproducible outcomes
- Lower variability between treatments
Low Exosome Counts
Some products contain significantly fewer exosomes per dose due to:
- Limited cell culture yield
- Dilution during formulation
- Inefficient isolation methods
In some cases, products are not standardized for particle count at all.
Cost advantage:
- Reduced production scale
- Faster processing
- Lower technical requirements
Clinical consequence:
- Suboptimal signaling to target cells
- Reduced biological effect
- Inconsistent clinical response
Presence of Contaminants
Purity is equally critical. Alongside exosomes, the production process may introduce:
- Free proteins and cytokines (non-vesicular)
- Cell debris and apoptotic bodies
- Residual culture media components
- Endotoxins (in poorly controlled systems)
Removing these impurities requires:
- Multi-step purification
- Advanced filtration or chromatography
- Rigorous quality control testing
Cost implication:
- Each additional purification step increases:
- Time
- Material loss
- Operational complexity
Clinical implication:
- High purity → targeted, controlled biological signaling
- Low purity → “noisy” or diluted signaling, potential inflammatory responses
The Dilution Problem (Hidden Cost Reduction Strategy)
One of the most common cost-reduction approaches is post-isolation dilution:
- A small amount of exosomes is mixed into a larger volume of carrier solution
- Final product appears similar in packaging, but contains lower active payload
This significantly reduces:
- Cost per unit
- Production burden
But also reduces:
- Biological potency
- Dose reliability
In exosome therapy, the effective dose is not defined by volume—but by the number of functional vesicles and their purity.
Two products with identical volume (e.g., 5 mL) may differ dramatically in Particle count, Functional cargo delivery, Clinical outcome.
4. Technology and R&D Investment
Beyond basic manufacturing, a major differentiator between exosome products lies in the level of technological development and research investment behind them. High-end exosome platforms are not simply “collected vesicles”—they are the result of engineered systems designed to optimize functionality, stability, and delivery efficiency.
Proprietary Isolation and Processing Technologies
Standard isolation methods (e.g., basic centrifugation or filtration) can recover exosomes, but often with:
- Lower purity
- Structural damage
- Inconsistent yield
Advanced manufacturers invest in proprietary technologies, such as:
- Tangential Flow Filtration (TFF)
- Size-exclusion chromatography (SEC)
- Multi-step purification platforms
These systems are designed to:
- Preserve membrane integrity
- Maintain functional protein and RNA cargo
- Achieve high purity with minimal contamination
Analogy:
This is similar to extracting a delicate molecule in pharmaceutical production.
A basic method is like using a rough filter—you get something usable, but mixed and partially damaged.
Advanced systems are like high-performance chromatography in drug manufacturing, where you isolate the exact compound in its most active form.
Cost implication:
- Specialized equipment
- Multi-step processing
- Highly trained personnel
Clinical implication:
- More intact and functional exosomes
- Greater biological activity
- Better consistency between treatments
Functional Enhancement and Bioengineering
Some advanced platforms go beyond natural exosomes and incorporate functional enhancement strategies.
These include:
- Peptide or growth factor integration
- Surface modification for targeting
- Hybrid delivery systems (e.g., HA-based carriers, liposomal systems)
- Matrix modulation technologies to improve penetration
Analogy:
Think of this like the difference between:
- A standard oral medication, where only part of the drug reaches the target
- And a targeted drug delivery system (like nanoparticle chemotherapy), designed to deliver the active compound precisely where it is needed
In the same way, engineered exosome systems are designed to:
- Increase delivery efficiency
- Improve cellular uptake
- Maximize biological effect per dose
Cost implication:
- Significant R&D investment
- Complex formulation and validation
- Advanced testing and optimization
Clinical implication:
- Higher bioavailability
- Stronger and faster response
- More efficient use of each dose
Stability and Preservation Technologies
Exosomes are biologically fragile and sensitive to:
- Temperature changes
- Enzymatic degradation
- Mechanical stress
Advanced platforms use:
- Optimized lyophilization (freeze-drying)
- Stabilizing buffers and cryoprotectants
- Controlled storage and reconstitution systems
Analogy:
This is comparable to vaccine preservation.
A poorly stabilized vaccine may lose effectiveness before administration, even if the active ingredient was initially present.
Similarly, exosomes without proper stabilization may lose their functional integrity before reaching the patient.
Cost implication:
- Additional processing steps
- Stability studies
- Specialized storage and logistics
Clinical implication:
- Reliable potency at the time of use
- Reduced variability due to degradation
The true value of an exosome product lies not only in what it contains—but in how effectively that biological signal is preserved and delivered.
5. Stability and Shelf-Life
Exosomes are biologically active nanostructures with a lipid bilayer membrane and sensitive molecular cargo (proteins, mRNA, miRNA).
As such, they are inherently unstable outside controlled conditions and highly susceptible to degradation. Maintaining stability is essential to ensure that the exosomes delivered at the point of use retain their structural integrity and biological function.
Challenges in Exosome Stability
Exosomes can be compromised by:
- Temperature fluctuations
- Membrane destabilization
- Loss of functional cargo
- Enzymatic degradation
- Breakdown of proteins and nucleic acids
- Mechanical stress
- Vesicle rupture during handling or transport
- Oxidative damage
- Lipid peroxidation affecting membrane integrity
Even if a product initially contains a high concentration of exosomes, improper stabilization can render them biologically inactive over time.
Advanced Stabilization Technologies
High-quality exosome products rely on specialized stabilization strategies, including:
- Optimized lyophilization (freeze-drying)
- Removes water while preserving vesicle structure
- Requires precise control of temperature and pressure
- Use of cryoprotectants and stabilizing agents
- Protect membrane integrity during freezing and drying
- Prevent aggregation or fusion of vesicles
- Controlled formulation environments
- pH optimization
- Ionic balance to maintain structural stability
- Validated storage protocols
- Defined temperature ranges (e.g., refrigerated or ultra-low storage)
- Protection from light and environmental stress
Analogy
This is comparable to biologic drugs or vaccines, where the active component is highly sensitive to environmental conditions.
A product may be potent at the time of manufacture, but without proper stabilization:
- Its activity can decline significantly before administration
- The delivered dose becomes functionally lower than expected
From a Manufacturing Perspective
- Requires additional processing steps (e.g., lyophilization cycles)
- Involves specialized materials (cryoprotectants, stabilizers)
- Demands stability testing under multiple conditions (real-time and accelerated)
- Requires controlled cold-chain logistics and validated packaging
Clinical Implication
- Stable products deliver consistent biological activity at the point of use
- Reduced variability between batches and treatment sessions
- Greater confidence in treatment outcomes
In contrast, poorly stabilized products may:
- Lose potency over time
- Deliver inconsistent or diminished results
- Compromise reproducibility in clinical practice
The effectiveness of an exosome product is not determined at the time of production—but at the moment it is applied to the patient.
The Impact of Quality on Clinical Outcomes
In regenerative medicine, quality is not a luxury—it is a necessity.
High-quality exosome products provide:
- Predictable and reproducible results
- Stronger biological response
- Greater patient satisfaction
Lower-quality alternatives may result in:
- Minimal or inconsistent effects
- Reduced practitioner confidence
- Difficulty in building long-term treatment protocols
Clinical Perspective
As exosome-based therapies continue to evolve, the market will inevitably become more competitive and more complex.
For clinicians and decision-makers, the key is not simply to ask:
“Which product is more affordable?”
But rather:
“Which product is scientifically reliable, consistently manufactured, and biologically effective?”
In a field driven by cellular communication and regenerative potential,
the integrity of the process defines the integrity of the result.




