HYBRID EXOSOME BLESKIN EXXO


SKIN SET
- Modulates inflammatory pathways (including NF-κB signaling)
- Enhances fibroblast proliferation and dermal activity
- Stimulates collagen type I synthesis
- Supports angiogenesis and microvascular function
- Reinforces epidermal barrier integrity
- Promotes extracellular matrix remodeling
- Accelerates post-procedural tissue recovery
- Improves dermal hydration via hyaluronan support
- Enhances dermal-epidermal adhesion (laminin & fibronectin support)
- Optimizes cellular turnover and regenerative signaling

HAIR SET
- Activates Wnt/β-catenin signaling pathway
- Promotes anagen phase initiation
- Enhances dermal papilla cell activity
- Supports perifollicular angiogenesis
- Modulates inflammatory mediators associated with miniaturization
- Improves follicular microenvironment homeostasis
- Stimulates extracellular matrix remodeling around follicular unit
- Enhances follicular stem cell niche signaling
- Supports shaft thickness and follicular anchoring
- Optimizes integration with PRP, microneedling, energy-based therapies
EXXO SKIN + PURISMA Clinical Application
Experience a youthful glow with our cutting-edge exosome solutions. Lactobacillus-formula strengthens the skin barrier and promotes regeneration, effectively slowing down the aging process.
WHAT IS BLESKIN EXXO EXOSOME?
EXXO SKIN BOOSTER
EXXO SKIN is a next-generation regenerative exosome formulation engineered to optimize dermal repair and cellular communication. Utilizing bioactive extracellular vesicles combined with Lactobacillus-derived postbiotic complexes, it helps modulate inflammatory pathways (including NF-κB signaling), reinforce epidermal barrier integrity, and restore cutaneous homeostasis following aesthetic procedures.
The formulation is enriched with high-purity adipose-derived stem cell conditioned media and PDRN (polydeoxyribonucleotide), supporting fibroblast proliferation, collagen type I synthesis, angiogenesis, and extracellular matrix remodeling. By enhancing cellular turnover and accelerating tissue recovery, EXXO SKIN is strategically positioned for anti-aging protocols, post-energy device recovery, barrier-compromised skin, and regenerative combination treatments.

Through its multi-pathway regenerative approach, EXXO SKIN enhances dermal-epidermal cross-talk and supports growth factor–mediated signaling, including pathways associated with TGF-β and VEGF regulation. This contributes to improved microvascular support, optimized nutrient diffusion, and more efficient extracellular matrix organization. The presence of structural matrix-associated proteins such as laminin, fibronectin, and hyaluronan further supports cellular adhesion, hydration balance, and tissue architecture stabilization. By addressing both inflammatory modulation and structural regeneration, EXXO SKIN provides aesthetic professionals with a biologically driven solution designed for predictable integration into advanced clinical protocols.
Patented Regenerative Bio-Complex
A Korean-patented bioactive complex integrating growth factor–associated signaling molecules and extracellular matrix components to support targeted cellular regeneration and tissue repair.











EXXO HAIR supports follicular longevity by influencing cellular energy metabolism and oxidative stress regulation within the scalp microenvironment. Through exosome-mediated signaling, it helps attenuate inflammatory mediators associated with follicular miniaturization and promotes a balanced cytokine profile conducive to sustained anagen maintenance. By reinforcing extracellular matrix integrity around the follicular unit and enhancing microvascular dynamics, EXXO HAIR contributes to improved shaft thickness, structural anchoring, and overall scalp resilience, making it suitable for structured, protocol-based hair restoration programs in advanced aesthetic practice.
EXXO HAIR BOOSTER
EXXO HAIR is a regenerative exosome formulation engineered to influence key molecular pathways governing follicular cycling, particularly the Wnt/β-catenin signaling pathway, a central regulator of hair follicle stem cell activation and anagen initiation. By delivering bioactive extracellular vesicles containing growth factors, peptides, and regulatory microRNAs, EXXO HAIR supports the stabilization of β-catenin within dermal papilla cells, promoting transcription of genes associated with follicular proliferation and differentiation.
Through modulation of perifollicular inflammation and enhancement of dermal papilla–epithelial cross-talk, the formulation helps create a pro-anagen microenvironment. The inclusion of adipose-derived stem cell conditioned media and PDRN further supports angiogenesis, fibroblast activation, and extracellular matrix remodeling — optimizing oxygenation and nutrient delivery to the follicular unit.
By targeting both signaling activation and microenvironment correction, EXXO HAIR is positioned as a biologically driven protocol for androgenetic alopecia, telogen effluvium, and early-stage follicular miniaturization, particularly in combination with microneedling, PRP, or energy-based scalp therapies.

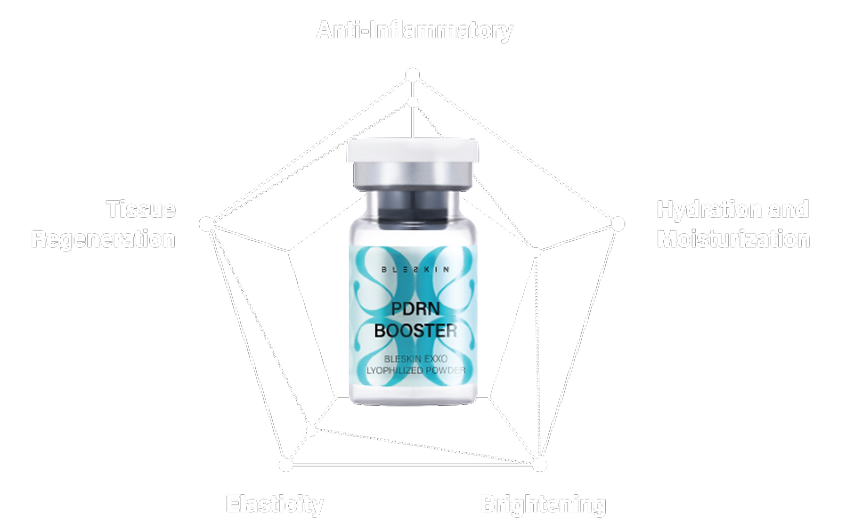
EXXO PDRN BOOSTER
This unique formulation combines hydrating, regenerating, and protective ingredients to create a versatile and effective skincare solution for anti-aging, brightening, and moisturizing. Revitalize your skin with advanced hydration and repair, powered by over 2% PDRN.
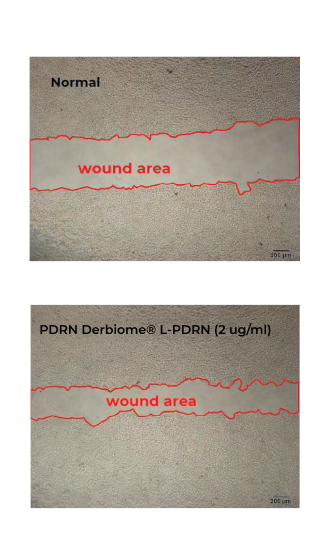
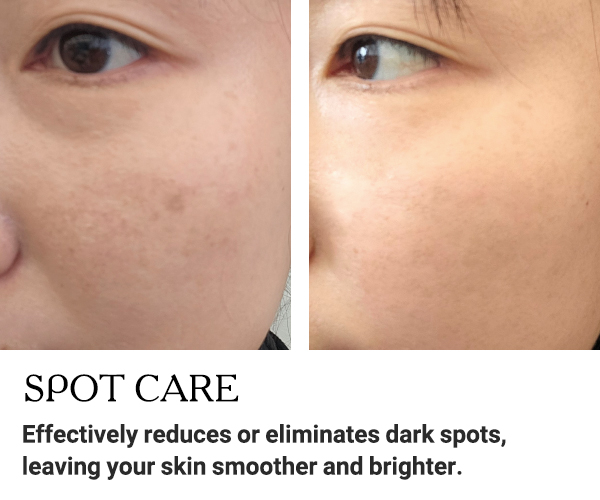
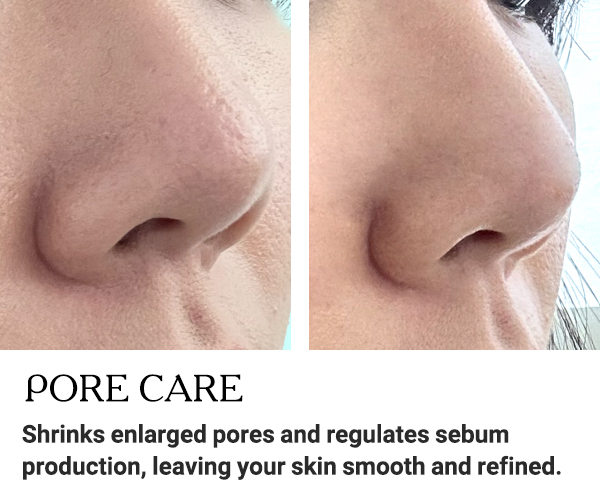
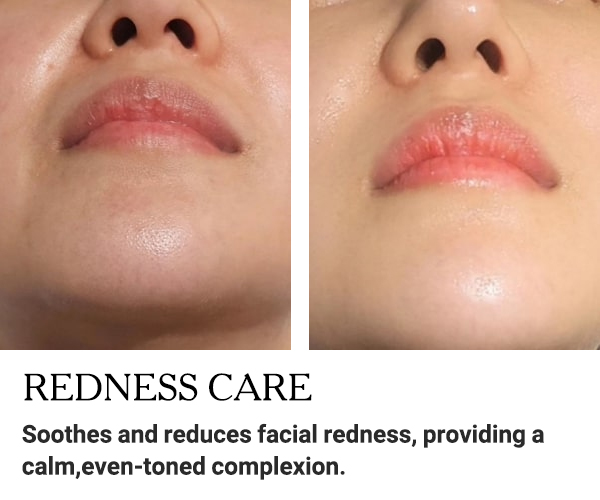
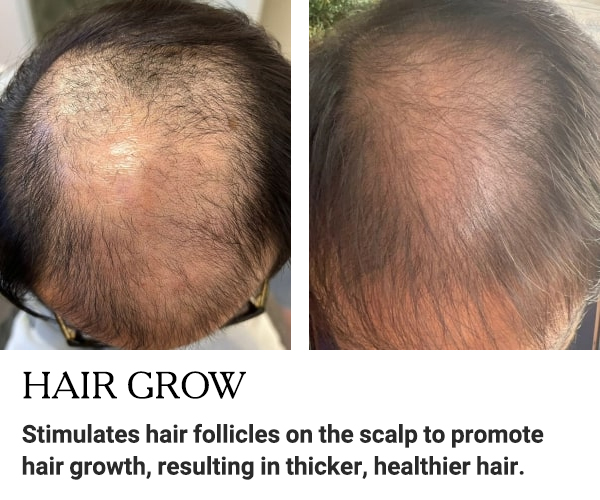
BEFORE AND AFTER RESULTS
Discover the amazing transformation with EXXO.
See the impressive before and after results!